SARS-CoV-2
RNA STAR Complete
LumiraDx SARS-CoV-2 RNA STAR Complete is a rapid, non-isothermal nucleic acid amplification qSTAR method intended for the qualitative detection of nucleic acid from SARS-CoV-2 in nasopharyngeal swabs collected from individuals suspected of COVID-19 by their healthcare provider (HCP).
- Reliable and robust performance when compared to traditional RT-PCR tests
- Direct amplification within 20 minutes drives lab's efficiency and productivity
- Based on in silico analysis we confirm detection of all known circulating variants including Omicron, Omicron B.2 and Delta
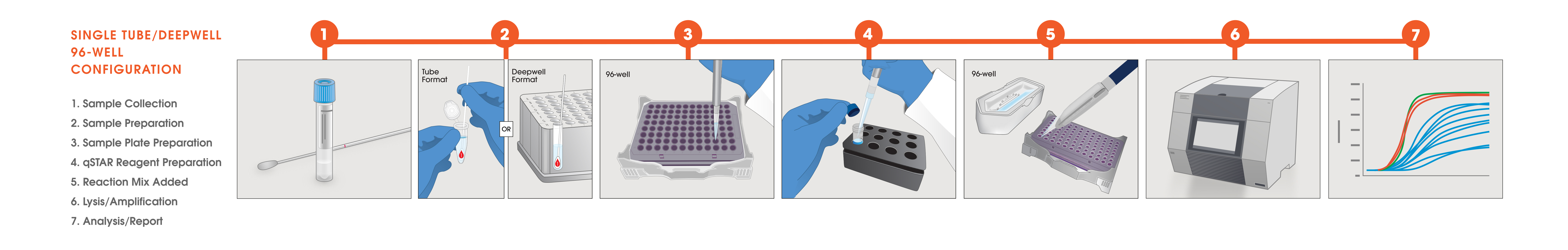
Test Workflows
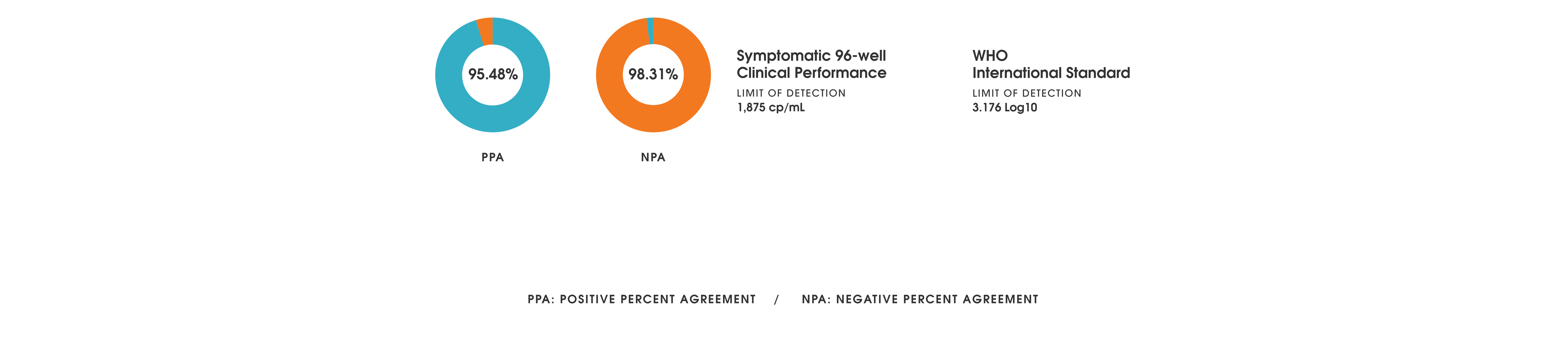
Performance
Join the Fast Lab Network

For more information about LumiraDx SARS-CoV-2 RNA STAR Complete:
LumiraDx SARS-CoV-2 RNA STAR reagents are for research use only and not for use in diagnostic procedures.
Not all products are available in all countries and regions. Please check with your local LumiraDx sales representative or distributor for availability in specific markets. Available in the US under FDA Emergency Use Authorization.