SARS-CoV-2 Ag Pool Test
a rapid microfluidic immunofluorescence assay designed to test 5 samples in 12 minutes. Used with the LumiraDx Platform, the LumiraDx SARS-CoV-2 Ag Pool test provides actionable, lab-comparable and cost efficient results for pooled patient samples in just 12 minutes.
Qualitative SARS-CoV-2 Ag Pool test
The LumiraDx SARS-CoV-2 Ag Pool test is an automated rapid microfluidic immunofluorescence assay for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 directly from nasal swab and nasopharyngeal swab samples collected from individuals suspected of COVID-19 or from asymptomatic individuals.
Near-patient testing
As an automated in vitro diagnostic test, it is made for near-patient testing with the LumiraDx Instrument.
Purpose
The test is a single use fluorescence immunoassay designed to detect the presence of the nucleocapsid protein antigen from SARS-CoV-2 in 1 to 5 swab samples.
Benefits

Rapidly identify COVID-19 in just 12 minutes
Verify potential infection and help guide infection control measures quickly:
- Save time: pooled testing can result in a 40 - 60% reduction in testing time and resources
- Sample type: nasal swab
- 100% PPA and 96.6% NPA when compared to LumiraDx SARS-CoV-2 Ag test
- Time to result: 12 minutes for up to 5 pooled samples
- Storage at room temperature (2°C to 30°C)

Workflow
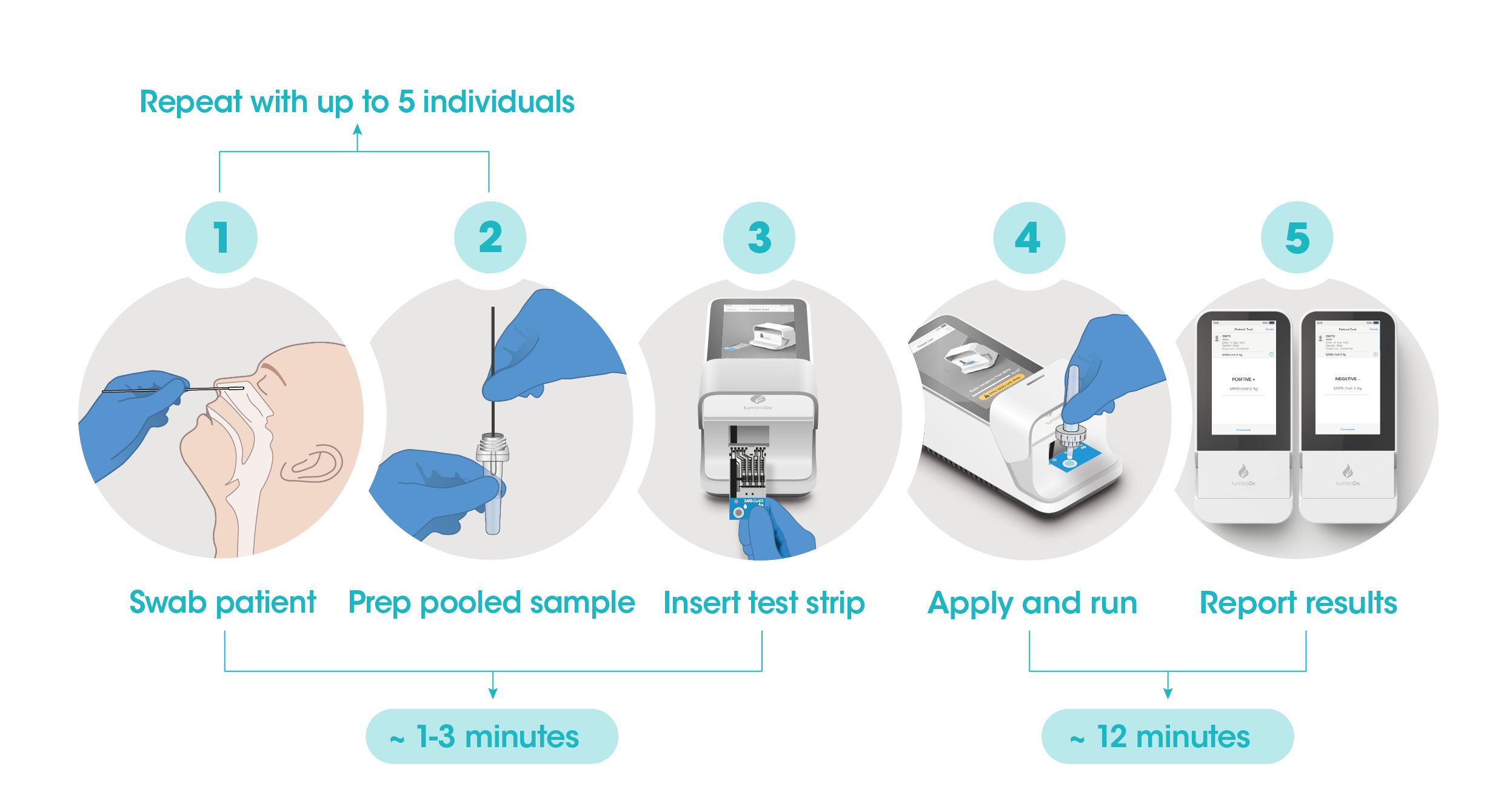
The workflow process is comprised of a simple sample prep along with step-by-step guidance of the Instrument to report a patient result in 12 minutes from sample application.
The Instrument and test strips are integrated with several quality control checks to ensure the Instrument and test are functioning correctly for every test run.
HOW TO USE VIDEOTest Performance
Clinical performance
In clinical studies, the LumiraDx SARS-CoV-2 antigen pool test demonstrated 100% positive agreement and 96.6% negative agreement when compared to testing individual swabs on the LumiraDx SARS-CoV-2 Ag test, thereby ensuring high sensitivity is maintained while reducing overall testing times, costs and operators required for testing.
Analytical performance
The limit of detection was found to be 32 TCID50/mL using a starting concentration of 2.8 x 105 TCID50/mL with 20/20 positive results.
Cross reactivity
SARS-CoV-2 Ag test was found not to cross-react with a panel of organisms and viruses including several human coronaviruses. See LumiraDx SARS-CoV-2 Ag test Product Insert for full details.
Product Documentation
SARS-CoV-2 Ag Pool Test:
SARS-CoV-2 Ag Pool Test

